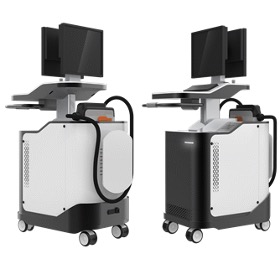
DOTTER Inc., a company that provides innovative cardiovascular solutions with advanced diagnostic technology, has completed product development using cardiovascular OCT imaging technology that can precisely observe the shape of lesions. This product is a Benetis Lifetime Model B and has obtained a permit (No. 23-4208) from MFDS in February 2023. Benetis Lifetime Model B is used with BentisTM (No. 21-4682), an MFDS-approved fiber optic catheter from Dotter. This optical imaging technique can obtain a three-dimensional image of the coronary artery microstructure. Compared to ultrasound (IVUS), the microstructure of normal and diseased vessels can be photographed more precisely.
Competitor company A has a resolution of 90kHz, while Dotter has developed equipment with a resolution of 200kHz and is equipped with superior performance and advanced technology compared to competitors.